The Cleanroom Grease Gun: A Maintenance Tool or an Audit Trap?
Anyone who has stepped into an ISO 5 fill-finish room during a lubrication task knows the feeling: cold air, a sudden pause in production, and that faint smell of grease that has no business near sterile drug products.
This Verdusco Automation article exposes the manual greasing risks hiding inside lyophilizer unloading systems and shows how a simple automation upgrade can protect your batches, your schedule, and your team.
Manual Greasing Risks That Quietly Put Your Line and Schedule on the Line
In a GMP area, the rule is simple: nothing is supposed to touch anything. Yet, manual greasing forces a collision between mechanical necessity and sterile integrity. We’ve all seen it. And, if we’re honest, we’ve grown used to it.
Think about the ritual:
The unloading sequence stops.
A technician spends twenty minutes gowning, only to bring a non-sterile grease gun into the heart of the line.
There’s the faint, metallic scent of industrial lubricant wafting near open syringes or vials, a smell that should never exist in a Class A zone.
Then comes the guesswork:
One “pump” too many, and you have grease weeping onto a conveyor.
One too few, and the bearing seizes mid-batch.
Still, the real friction happens later.
During an audit, QA asks, “Was this lubricated during the batch? Show me the log.”
You’re left relying on a handwritten note scribbled after the fact, or worse, the technician’s memory.
Manual greasing risks aren’t just about a drip of oil. They are about half a day lost to resterilization, the tension of a looming deviation report, and the high cost of human proximity to a sterile path.
If this scene makes you uneasy, you’re not overreacting. It’s your process telling you it’s time to change.
A Cleaner Way to Grease Equipment Without Entering the Room: Condition‑Based Lubrication in GMP Areas
Automation doesn’t have to be a “moonshot” project. It is often most effective when it solves a messy, repetitive problem with a quiet, invisible solution. Imagine a system where the “grease gun” stays outside the cleanroom entirely.
By implementing condition-based lubrication in GMP areas, you install a closed-loop system consisting of a central pump, located in a mechanical gray space or a lower-classified room. The pump delivers precise amounts of lubricant through sealed, high-pressure lines straight to the bearings and slides of your lyo-unloader.
The benefits are immediate and visceral:
No human entry into the sterile zone for lubrication. No gowning. No tools. No proximity to exposed vials.
Controlled delivery, triggered by cycle counts or operating conditions, not guesswork.
Automatic documentation, with time-stamped, tamper-proof records ready for batch review or audit.
You move from hoping lubrication was done correctly to knowing it was, without interrupting production or compromising sterility.
Nothing dramatic has changed on the floor. And that’s the point.
The ROI of Removing Manual Greasing From GMP Areas
Automation conversations often drift into “technology for technology’s sake.” Plant leaders don’t have time for that. They see cost, risk, and accountability.
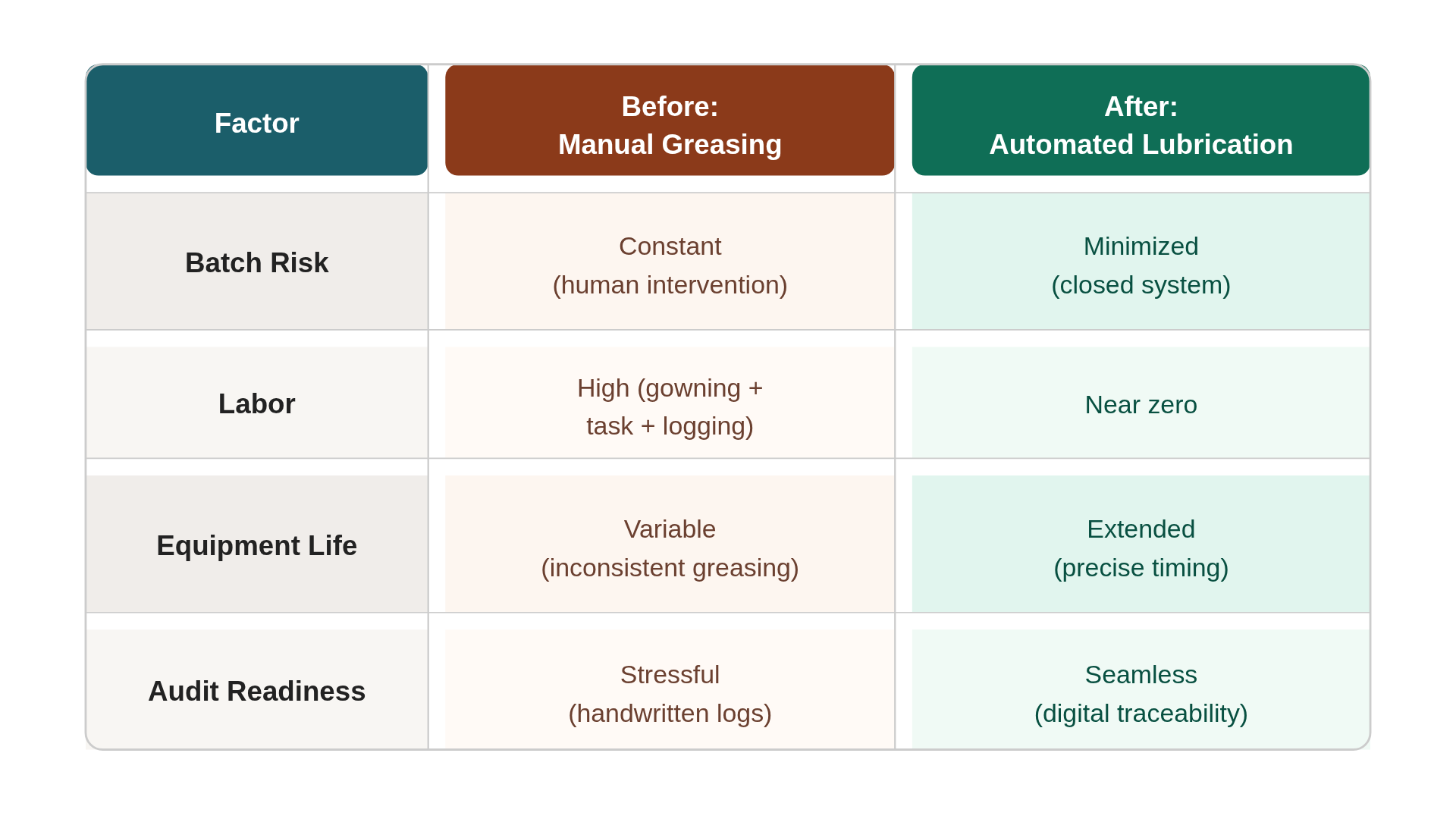
Consider the “Before” state:
Two hours of cumulative downtime per week for lubrication and gowning, plus the $250,000 “ghost cost” of a potential batch loss due to a contamination event. Industry benchmarks indicate that companies adopting automated maintenance see a significant reduction in unplanned downtime, often by 20% to 30%.
Here’s a before vs. after snapshot:
The “After” state is one of quiet, predictable flow. You aren't fighting fires or scrubbing grease off a conveyor belt. You’re simply running a cleaner, more profitable line.
Your Next Step: Eliminate the Grease Trap With a Solid Partner
Change doesn’t have to be loud. Sometimes, it just means removing the grease gun from the room and never looking back.
If you’re considering this shift, you don’t need a blank-check budget or a year-long project. You need clarity and a partner who understands GMP reality.
At Verdusco Automation, we help manufacturers adopt automation in ways that feel achievable, not overwhelming. For challenges like manual greasing risks in aseptic areas, we support you through:
Industrial automation design tailored to existing lyophilizer unloading systems.
Control systems integration that enables condition-based lubrication and clean data logging.
Integrated automation solutions that fit GMP environments without disrupting operations
The result is safer processes, cleaner audits, and teams that spend less time firefighting and more time running the plant.
Getting started is easy.
Just contact Verdusco Automation today, using your preferred channel:
➔ email: maria@verduscoautomation.com
➔ LinkedIn: https://www.linkedin.com/in/raul-automation/
➔ website: https://www.verduscoautomation.com/contact
______
Final note: This article shows what’s possible. A precise ROI and design always start with a quick, custom look at your facility. We make that step straightforward and low-pressure.